Thermotoga
maritima glucosidase A (AglA): A glucosidase with an NAD+ cofactor
|
Family 4
glycoside hydrolases (GH4) represent an unusual group of glucosidases with
a requirement for NAD+, divalent metal cations and reducing conditions.
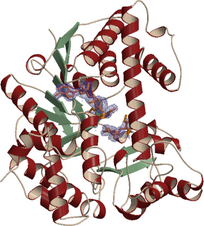
AglA from Thermotoga maritima is a typical GH4 enzyme, requiring NAD+, Mn2+
and strongly reducing conditions for activity. We have determined the crystal
structure of the protein complexed with NAD+ and maltose refined at 1.9 Å
resolution. The NAD+ is bound to a typical Rossman-fold NAD+-binding site
and the nicotinamide moiety is localized close to the maltose substrate.
Within the active site the conserved Cys174 and surrounding histidines are
positioned to play a role in the hydrolysis reaction. The electron density
maps indicate that Cys174 is oxidized to a sulfinic acid. Most likely, the
strongly reducing conditions are necessary to reduce the oxidised cysteine
side chain. Notably, the canonical set of catalytic acidic residues common
to other glucosidases is not present in the active site. This, combined with
a high structural homology to NAD-dependent dehydrogenases suggests an unusual
and possibly unique mechanism of action for a glycoside-hydrolysing enzyme.
|
Reference
J.A.
Lodge, T. Maier, W.
Liebl, V. Hoffmann and N. Sträter
Crystal structure of Thermotoga
maritima alpha-glucosidase
AglA defines a new clan of NAD+-dependent
glycosidases
J. Biol. Chem. 2003,
278,
19151-19158
|



