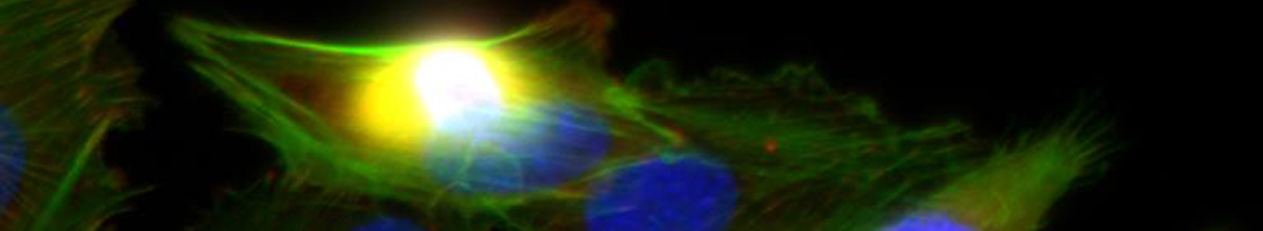
B10 – In situ characterization of mediator controlled immune cell interaction in GAG-modified aECM
The project aims on the mechanistic characterization of cellular interactions of macrophages and fibroblasts in three-dimensional fibrillar biomaterials with defined mechanical properties and GAG-modifications in vitro. In particular we investigate the modulation of paracrine cell-cell interactions via relevant mediators (IL-10, TGF-beta, MCP-1) by the GAG modification of the collagen based matrices. Besides quantitative analysis of transport kinetics of mediators in the fibrillar matrices, we characterize proliferation, migration and differentiation on single cell level and in a time-resolved manner of the coculture systems.
Publications
- Ullm F, Pompe T. Fibrillar biopolymer-based scaffolds to study macrophage-fibroblast crosstalk in wound repair. Biological Chemistry, vol. 402, no. 11, 2021, pp. 1309-1324. https://doi.org/10.1515/hsz-2021-0164.
- Vogel S, Ullm F, Müller CD, Pompe T, Hempel U. Impact of binding mode of low-sulfated hyaluronan to 3D collagen matrices on its osteoinductive effect for human bone marrow stromal cells. Biological Chemistry, vol. 402, no. 11, 2021, pp. 1465-1478. https://doi.org/10.1515/hsz-2021-0212.
- Logie C, Schaik T, Pompe T, Pietsch K. Fibronectin-functionalization of 3D collagen networks supports immune tolerance and inflammation suppression in human monocyte-derived macrophages. Biomaterial Volume 268, January 2021. https://doi.org/10.1016/j.biomaterials.2020.120498
- Ansorge M, Pompe T. Systems for localized release to mimic paracrine cell communication in vitro. J Control Release. 2018; 278:24-36.
- Müller C, Ansorge M, Espig M, Zschoche S, Schiller J, Pompe T: Covalent binding of maleic anhydride copolymer monolayers to polyacrylamide hydrogels. Macromol Chem Phys. 2018.
- Sapudom J, Kalbitzer L, Wu X, Martin S, Kroy K, Pompe T. Fibril bending stiffness of 3D collagen matrices instructs spreading and clustering of invasive and non-invasive breast cancer cells. Biomaterials. 2018; 193:47-57.
- Ansorge M, Sapudom J, Chkolnikov M, Wilde M, Anderegg U, Möller S, Schnabelrauch M, Pompe T. Mimicking paracrine TGF-ß1 signals during myofibroblast differentiation in 3D collagen networks. Scientific Reports. 2017, 7:5664.
- Sapudom J, Wu X, Chkolnikov M, Ansorge M, Anderegg U, Pompe T. Fibroblast fate regulation by time dependent TGF-ß1 and IL-10 stimulation in biomimetic 3D matrices. Biomater Sci. 2017, 5(9):1858-1867.
- Friedemann M, Kalbitzer L, Franz S, Moeller S, Schnabelrauch M, Simon JC, Pompe T, Franke K. Instructing human macrophage polarization by stiffness and glycosaminoglycan functionalization in 3D collagen networks. Adv Healthc Mater. 2017, 6. DOI: 10.1002/adhm.201600967
- Sapudom J, Ullm F, Martin S, Kalbitzer L, Naab J, Möller S, Schnabelrauch M, Anderegg U, Schmidt S, Pompe T. Molecular weight specific impact of soluble and immobilized hyaluronan on CD44 expressing melanoma cells in 3D collagen matrices. J Actbio 2016; DOI: 10.1016/j.actbio.2016.12.026.
- Martin S, Wang H, Rathke T, Anderegg U, Möller S, Schnabelrauch M, Pompe T, Schmidt S. Polymer hydrogel particles as biocompatible AFM probes to study CD44/hyaluronic acid interactions on cells. Polymer. 2016;102:342–349.
- Sapudom J, Rubner S, Martin S, Pompe T. Mimicking tissue boundaries by discrete multi-parameter matrix interfaces. Adv Healthcare Mat. 2016;5:1861-7.
- Ansorge M, Rastig N, Steinborn R, König T, Baumann L, Moller S, Schnabelrauch M, Cross M, Werner C, Beck-Sickinger AG, Pompe T. Short-range cytokine gradients to mimic paracrine cell interactions in vitro. J Control Release. 2016;224:59-68.
- Kalbitzer L, Franke K, Möller S, Schnabelrauch M, Pompe T. Glycosaminoglycan functionali-zation of mechanically and topologically defined collagen I matrices. J Materials Chemistry B. 2015;3:8902-10.
- Sapudom J, Rubner S, Martin S, Thoenes S, Anderegg U, Pompe T. The interplay of fibronectin functionalization and TGF-beta1 presence on fibroblast proliferation, differentiation and migration in 3D matrices. Biomater Sci. 2015;3:1291-301.
- Franke K, Sapudom J, Kalbitzer L, Anderegg U, Pompe T. Topologically defined composites of collagen types I and V as in vitro cell culture scaffolds. Acta Biomater. 2014 Jun;10(6):2693-702. doi: 10.1016/j.actbio.2014.02.036. Epub 2014 Mar 1.
- Stamov DR, Pompe T. Structure and Function of ECM-inspired composite collagen I scaffolds. Soft Matter. 2012; 8: 10200-12.
- Müller C, Stamov DR, Werner C, Pompe T. Nanoscale Characterization of Cell Receptors and Binding Sites on Cell-Derived Extracellular Matrices. Ultramicroscopy. 2012; 118: 44-52.
- Teichmann J, Morgenstern A, Seebach J, Schnittler HJ, Werner C, Pompe T. The control of endothelial cell adhesion and migration by shear stress and matrix-substrate anchorage. Biomaterials. 2012; 33: 1959-69.
- Scherf N, Franke K, Glauche I, Kurth I, Bornhäuser M, Werner C, Pompe T, Roeder I. On the symmetry of siblings: automated single-cell tracking to quantify the behavior of hematopoietic stem cells in a biomimetic setup. Exp Hematol. 2012; 40: 119-30.
- Stamov DR, Khoa Nguyen TA, Evans HM, Pfohl T, Werner C, Pompe T. The impact of heparin intercalation at specific binding sites in telopeptide-free collagen type I fibrils. Biomaterials. 2011; 32: 7444-53.
- Pompe T, Kaufmann M, Kasimir M, Johne S, Glorius S, Renner L, Bobeth M, Pompe W, Werner C. Friction controled traction forces in cell adhesion. Biophys J. 2011; 101: 1863-70.
- Herklotz M, Werner C, Pompe T. The impact of primary and secondary ligand coupling on extracellular matrix characteristics and formation of endothelial capillaries. Biomaterials. 2008; 30: 35-44.
- Alberti K, Davey RE, Onishi K, George S, Salchert K, Seib FP, Bornhauser M, Pompe T, Nagy A, Werner C, Zandstra PW. Functional immobilization of signaling proteins enables control of stem cell fate. Nat Methods. 2008; 5: 645-50.
Contact

Prof. Dr. rer.nat. Tilo Pompe
Chair of Biophysical Chemistry
Leipzig University
Faculty of Life Sciences
Institute of Biochemistry
Johannisallee 21 – 23, 04103 Leipzig
Phone: +49 (0)341 97 36931
E-Mail: tilo.pompe@uni-leipzig.de
Web: biochemie.lw.uni-leipzig.de/arbeitsgruppen/biophysikalische-chemie/
